Compliance vs. Quality – Interphex Edition
By: Daniel R. Matlis, President
Last month, I had the privilege to hea r Lawrence Yu, PhD, FDA Deputy Director of CDER’s Office of Pharmaceutical Quality (OPQ) at INTERPHEX 2018.
r Lawrence Yu, PhD, FDA Deputy Director of CDER’s Office of Pharmaceutical Quality (OPQ) at INTERPHEX 2018.
The theme of his presentation “The Future of Pharmaceutical Quality and the Path to Get There,” was that consumers and patients deserve six sigma quality products.
To encourage this effort, the Agency began the drive to modernize its regulation of the pharmaceutical industry to emphasize Quality more than a decade ago with the 21st Century Initiative.
The vision, as was conveyed by Dr. Janet Woodcock Director, Center for Drug Evaluation and Research, is: “A maximally efficient, agile, flexible pharmaceutical manufacturing sector that reliably produces high quality drugs without extensive regulatory oversight.” –
This journey is still on-going with the agency working to ensure Quality is the underpinning of everything CDER does. During his session, Dr. Yu defined the criteria for a High Quality Product as follows – a product that:
- Reproducibly delivers the therapeutic benefit to the consumer as stated in the label
- Is free of defects
- Presents no undeclared risk (e.g., isn’t contaminated)
The Agency’s paradigm shift to drive the industry to focus on Quality, with Compliance as a baseline, is not a new concept to LSP readers. We started writing about this 12 years ago. To this end, FDA is shifting Pharmaceutical regulation to focus on improved performance of individual and organizational behavior in ways that protect and promote public health.
But is all this talk about quality falling on deaf ears? In my experience, there is significant regulatory inertia in most organizations impeding the shift from compliance to quality. Many quality departments are driven to meet regulatory compliance, thereby focusing on measuring the wrong metric.
Case in point, how does your quality organization measure the success of regulatory interactions? I bet it is inversely proportional to the number of 483s received.
How many Pharma quality departments measure quality in Sigmas? According to FDA and data obtained by Axendia, the agency estimates that the pharmaceutical industry hovers between 2 Ϭ and 3Ϭ resulting in a cost of quality around 25%. In contrast, the semiconductor industry consistently achieves 6Ϭ, which results in a cost of quality ranging from 1 to 3%.

Another factor hindering the shift is the market reward of Innovation over Quality. While quality issues may be a blip on a company’s stock price or a line item on a balance sheet, failure of a new compound in late stage development will cause valuations to tumble.
During a recent keynote, I emphasized the need for Life-Science Organizations to transition their focus from mere Compliance towards a culture of driving Quality improvements across the organization.
To incentivize industry, the agency is shifting to Performance-based Regulation. This regulatory approach focuses on desired, measurable outcomes, rather than prescriptive processes, techniques, or procedures. Performance-based regulation leads to defined results without specific direction regarding how those results are to be obtained.
Compliance is a Baseline, it is the cost of doing business in the Life-Sciences Industry. Collecting documented evidence of compliance with regulatory requirements is an overhead cost for Life Science companies.
By contrast, Quality is an Investment. Improvements in quality drive enhanced and consistent product performance. In addition streamlined processes result in operational efficiencies and reduce waste. Investing in improving product quality lowers costs and improves outcomes.
To support this transition, the agency has been steadily increasing the use of advanced machine learning and big data to push regulation to a performance-based model that emphasized metrics and performance capabilities versus compliance “box checking.”
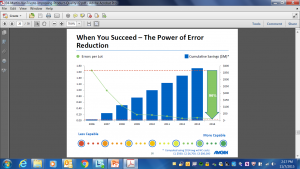 The use of process capability to ensure product quality has proven benefits. Amgen reduced errors per lot by 96% resulting in savings of ~$400 million. (Source: USFDA Future of Pharmaceutical Quality and the Path to Get There, Lawrence Yu, Ph.D. Deputy Director, Office of Pharmaceutical Quality FDA Center for Drug Evaluation and Research INTERPHEX 2018, April 18, 2018, Javits Center, NYC)
The use of process capability to ensure product quality has proven benefits. Amgen reduced errors per lot by 96% resulting in savings of ~$400 million. (Source: USFDA Future of Pharmaceutical Quality and the Path to Get There, Lawrence Yu, Ph.D. Deputy Director, Office of Pharmaceutical Quality FDA Center for Drug Evaluation and Research INTERPHEX 2018, April 18, 2018, Javits Center, NYC)
 At Lilly, a sustained focus on quality risk management has delivered measurable Improvements in Product Quality including a 90% reduction on backlog, and a 50% reduction in deviations. (Source: USFDA Future of Pharmaceutical Quality and the Path to Get There, Lawrence Yu, Ph.D. Deputy Director, Office of Pharmaceutical Quality FDA Center for Drug Evaluation and Research INTERPHEX 2018, April 18, 2018, Javits Center, NYC)
At Lilly, a sustained focus on quality risk management has delivered measurable Improvements in Product Quality including a 90% reduction on backlog, and a 50% reduction in deviations. (Source: USFDA Future of Pharmaceutical Quality and the Path to Get There, Lawrence Yu, Ph.D. Deputy Director, Office of Pharmaceutical Quality FDA Center for Drug Evaluation and Research INTERPHEX 2018, April 18, 2018, Javits Center, NYC)
Dr. Yu concluded his presentation with the following message:
Consumers and patients deserve six sigma quality products with minimal risks of shortages or recalls.
- The market needs to observe and reward Quality
- FDA oversight must move to performance-based Regulation
- Industry must develop and adopt emerging technologies
- Industry should adopt Quality by Design
- Industry needs to focus on continuous improvement and operational excellence
Is your organization ready to transition its focus from measuring NO 483s to surpassing 6Ϭ?
We can help you build a culture of quality and innovation.
Related reading:
Compliance vs Quality – India Edition
Compliance First Approach Still an Obstacle to Product Quality

